As of December 2023, 10 FDA-approved gene therapies were available. Based on existing development programs, 30-50 additional gene therapies are expected to be FDA-approved by 2030. The impact on treatment outcomes, but also the pressure on the budgets of payers, is already powerful. How does the uptake look in some of the countries of Central and Eastern Europe?
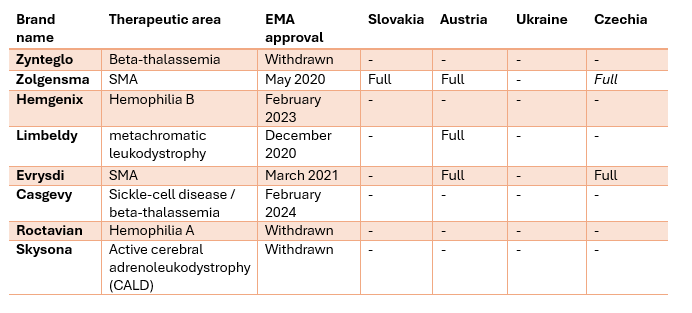
Table 1: Reimbursement status in Slovakia, Austria, Ukraine and Czechia of a set of eight gene therapy brands initially EMA-approved between 2019 and 2024.
Austria reimburses 3 of these in full, 1 each is officially fully reimbursed in Slovakia and Czechia, whilst Zolgensma is not in the official catalogue in Czechia, based on a special agreement between the largest health insurance company, VZP and the drug manufacturer Novartis, VZP has agreed to reimburse the drug. Notably, 3 of the 8 drugs (Zynteglo, Skysona and Roctavian) in our sample have been withdrawn from the EU market by the manufacturers themselves.
Get in touch for more info: info@ambiom.com
Sources:
(1) European Medicines Agency. Medicines. https://www.ema.europa.eu/en/medicines
(2) Všeobecná zdravotní pojišťovna ČR. “VZP umožní léčbu všem dětem s SMA indikovaným k léčbě přípravkem Zolgensma.” VZP ČR Aktuality, 16 Dec 2020. https://www.vzp.cz/o-nas/aktuality/vzp-umozni-lecbu-vsem-detem-s-sma-indikovanym-k-lecbe-pripravkem-zolgensma
(3) Ministerstvo zdravotníctva Slovenskej republiky. “Vitajte na portáli Kategorizácia!” Portál Kategorizácia (Registrácia používateľa). https://kategorizacia.mzsr.sk/
(4) Dachverband der Sozialversicherungsträger. ÖKO-Views. https://www.sozialversicherung.at/oeko/views/index.xhtml
(5) Státní ústav pro kontrolu léčiv (SÚKL). Seznam cen a úhrad LP/PZLÚ k 1. 5. 2024. https://www.sukl.cz/sukl/seznam-cen-a-uhrad-lp-pzlu-k-1-5-2024
No comments.