Clinical research success rates of different disease areas have far-reaching implications in the structure of corporate R&D budgets, pharmaceutical asset valuation and, eventually, M&A activity. The report published in February 2021 considers success rates of clinical drug development overall as well as by phase of clinical research over the past decade.
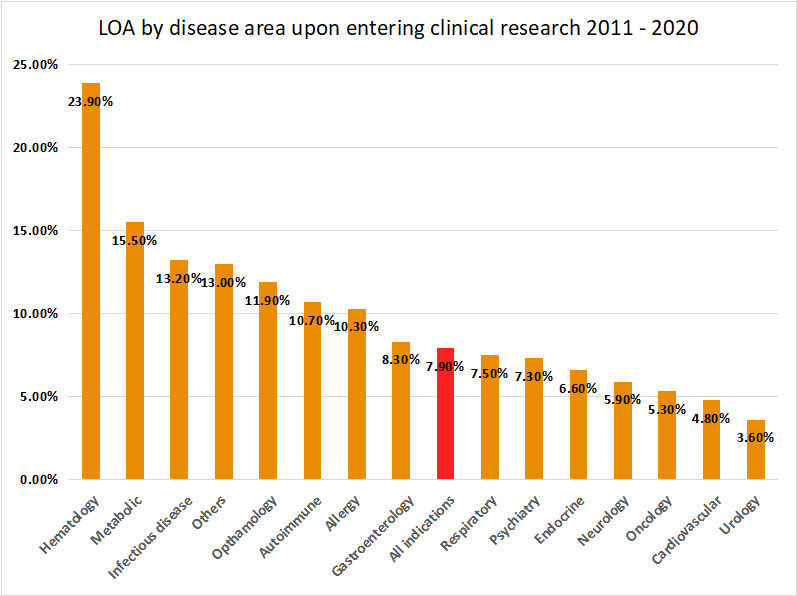
The data is comprised of lead as well as secondary indications. Across all indications, the likelihood of approval (LOA) from 2011 to 2020 was 7.9%. The clear outlier was the disease area of haematology, with almost a 1 in 4 chance of a drug being approved upon the start of clinical research. At the other end of the spectrum, urology, cardiovascular diseases and oncology disease areas have had the lowest approval rates with 3.6%, 4.8% and 5.3% respectively. According to the report, Phase II development remains the largest hurdle in clinical drug development, with just 28.9% of candidates achieving this critical phase transition.
Key takeaways for ambiom:
- From our point of view, it will be especially intriguing to observe the progress of LOA of the oncology disease area as novel immuno-oncology therapies boasted an LOA of 12.4% - more than twice the average for oncology overall.
- Notably, rare disease therapies have reached an overall LOA of 17.0%. We expect that this fact, coupled with a generally favourable rare disease reimbursement landscape in Europe and elsewhere, could propel further investment in rare disease drug discovery and development programmes.
To find out more about how we can help out with your pharma projects, reach out at info@ambiom.com.
Source:
Biotechnology Innovation Organization, Informa Pharma Intelligence, & QLS Advisors. (2021). Clinical Development Success Rates and Contributing Factors 2011–2020. Informa Pharma Intelligence. https://pharmaintelligence.informa.com/
Comments are closed.