Latest in: clinical research
Recently, the FDA has requested Sarepta Therapeutics, Inc., a Massachusetts-based biopharmaceutical company, to suspend all distribution of its DMD gene therapy Elevidys (delandistrogene moxeparvovec), following the deaths of three patients—two non‑ambulatory teenagers and one …
Introduction If you asked us (and many other industrial consultants) what one of the most common commercial strategy mistakes we see in otherwise well-positioned early-stage companies is, without a doubt, it would be the …
Drug development is a complex and resource-intensive process that involves significant investments in research, clinical trials, and regulatory activities. As pharmaceutical companies evaluate the potential of assets, they employ various financial metrics. Two commonly …
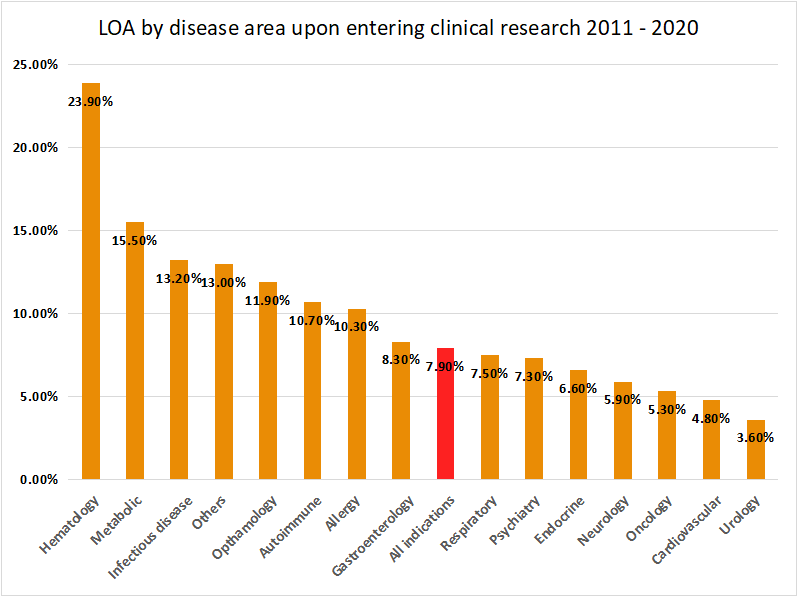
Clinical research success rates of different disease areas have far-reaching implications in the structure of corporate R&D budgets, pharmaceutical asset valuation and, eventually, M&A activity. The report published in February 2021 considers success rates of clinical …