Four years ago, we have produced what seemed to be one of our most resonant pieces of content, on the status of companion diagnostics industry in 2022. Over the past 4 years, the industry has come a long way, and we are convinced there is a time for a quick update on the current trends.
What do the approval numbers look like?
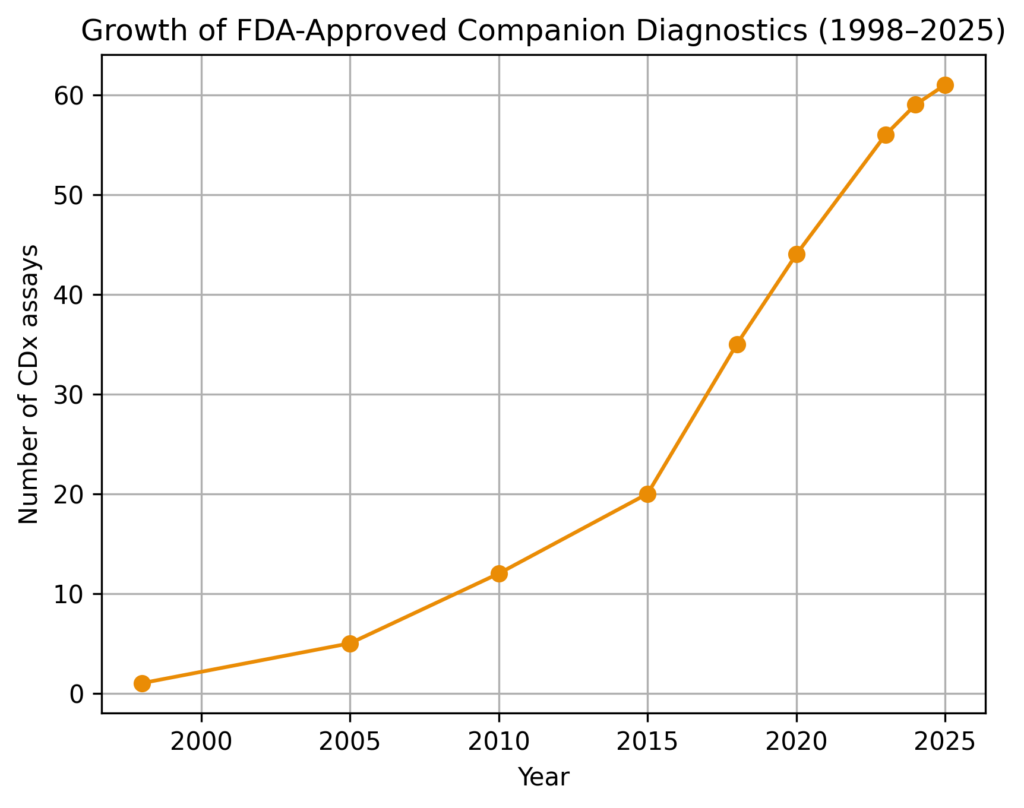
As of April 2022, 50 unique CDx assays have been approved, overwhelmingly for oncology indications. This has grown to 61 unique CDx assays as of late 2025. While oncology still dominates (~90%), non-oncology companion diagnostics have expanded to ~10% of approvals, driven almost exclusively by gene therapies, rare genetic diseases, and specialized hematology indications—with no meaningful penetration into common chronic diseases yet. There have been also some signs of discontinuation of availability of certain products, specifically of the POMC/PCSK1/LEPR CDx Panel.
Significantly, in late 2023, a broad-spectrum FoundationOne CDx (F1CDx) has been approved with expanded indications. FoundationOne CDx is an NGS-based assay designed to detect genetic variations in 324 genes in addition to select gene rearrangements and other biomarkers in the genomic makeup of a tumor. FoundationOne CDx is clinically and analytically validated for a whole spectrum of solid tumours and can provide corresponding clinical guidance on a spectrum of treatment options. We expect the trend of broad-spectrum CDx testing to continue, particularly based on the NGS platforms.
Current issues
Companion diagnostic regulations are intended to protect patients by ensuring the quality and consistency of treatment-guiding biomarker testing in clinical trials and clinical practice. However, the industry often reports that current regulations (e.g. CDx assays falling under the highest risk classes within IVDR - https://ambiom.com/companion-diagnostics-status-in-2022-and-future-outlook) have had unintended negative consequences relating to innovation, implementation, and accessibility of precision medicine; increasing complexity and cost burden; and inhibiting development of novel diagnostics and biomarker-targeted therapeutics.
In many instances, running a companion diagnostic assay is costly, raising questions about the capability of patients to pay and reimbursement. Indeed, slower uptake and lack of reimbursement are important challenges and often great roadblocks to clinical adoption, also in cases where the clinical and health economic benefit of pre-prescription testing is clear (https://ambiom.com/ngs-in-clinical-routine-where-are-we-in-2022).
In practice, CDx testing has often been done via laboratory-developed tests (LDT). However, the FDA has recently increased LDT oversight due to concerns that LDTs may not be as robust in performance as the approved companion diagnostic, although past real-world evidence showed very good results, particularly for single-mutation and next-generation sequencing LDTs. In the European Union, LDTs used in clinical decision-making must comply with current IVDR provisions and are effectively unsustainable.
Finally, inter-country, inter-lab variability, and variability driven by the selection of technology platforms remain significant concerns when assessing the predictive power of companion diagnostic assays.
Outlook
We reiterate our prognosis on the CDx space being dominated by oncology-focused assays, although it is clear that predictive testing has now started to spread to other indications. Technology-wise, NGS continues to be the method of choice for most of the new approved assays. Regulation, or lack thereof, is going to have a significant impact both in the US and EU on how the assays are developed, treated and whether or not they will stand a chance to continue to prove their cost- and clinical effectiveness in front of the reimbursement bodies.
Get in touch to learn more about how we could be helpful CDx-wise at info@ambiom.com.
Sources:
(1) Oliner, K. S., Shiller, M., Schmid, P., Ratcliffe, M. J., Schetter, A. J., & Tsao, M.-S. (2025). Challenges to innovation arising from current companion diagnostic regulations and suggestions for improvements. Clinical Cancer Research, 31(5), 795–800. https://pubmed.ncbi.nlm.nih.gov/39724199/
(2) PreventionGenetics. (2025, October 17). POMC/PCSK1/LEPR CDx panel discontinuation. https://www.preventiongenetics.com/pomc-pcsk1-lepr-cdx-panel-discontinuation
(3) U.S. Food and Drug Administration. (2025, March 5). List of cleared or approved companion diagnostic devices (in vitro and imaging tools). https://www.fda.gov/medical-devices/in-vitro-diagnostics/list-cleared-or-approved-companion-diagnostic-devices-in-vitro-and-imaging-tools
(4) U.S. Food and Drug Administration. (2024, June 18). FoundationOne CDx (F1CDx) – P170019/S048. https://www.fda.gov/medical-devices/recently-approved-devices/foundationone-cdx-f1cdx-p170019s048
No comments.