Latest in: market access
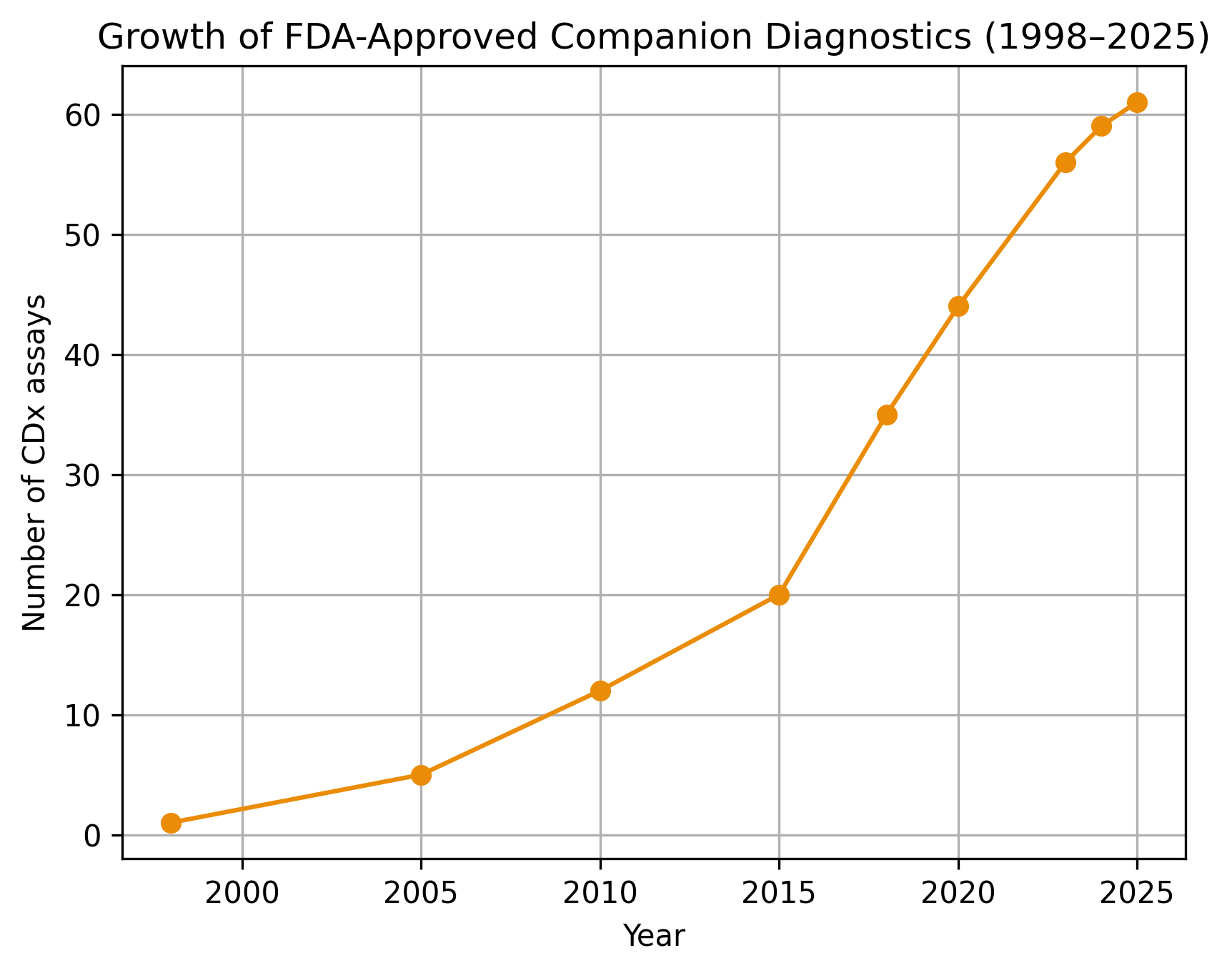
Four years ago, we have produced what seemed to be one of our most resonant pieces of content, on the status of companion diagnostics industry in 2022. Over the past 4 years, the industry …
Paradoxically, in a place not exactly known for its digital legacy, in 2019, a major change in the approach towards the reimbursement of digital health solutions has been put forward. The German Digital Healthcare …
Recently, the FDA has requested Sarepta Therapeutics, Inc., a Massachusetts-based biopharmaceutical company, to suspend all distribution of its DMD gene therapy Elevidys (delandistrogene moxeparvovec), following the deaths of three patients—two non‑ambulatory teenagers and one …
Introduction If you asked us (and many other industrial consultants) what one of the most common commercial strategy mistakes we see in otherwise well-positioned early-stage companies is, without a doubt, it would be the …
We spoke in detail about our activities at BeyondBiotech podcast, you can listen to the episode here: https://podcast.labiotech.eu/1995493/episodes/16219760-cracking-the-code-of-biotech-valuations
The new managed entry agreement (MEA) and health technology assessment (HTA) regulations adopted in Ukraine over the course of 2020 and 2021 present a significant step forward in improving access to treatment in the …