Latest in: gene therapy
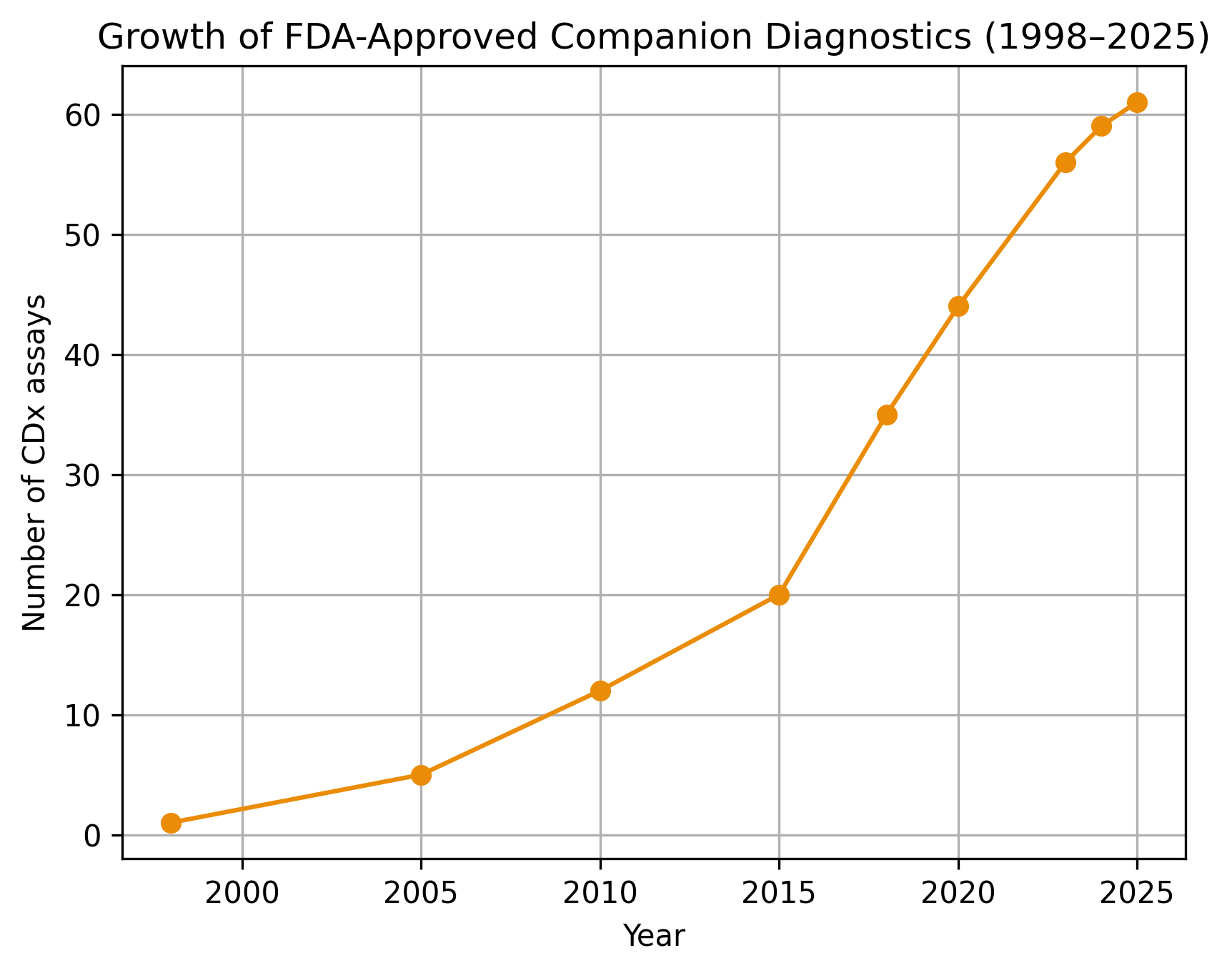
Four years ago, we have produced what seemed to be one of our most resonant pieces of content, on the status of companion diagnostics industry in 2022. Over the past 4 years, the industry …
Recently, the FDA has requested Sarepta Therapeutics, Inc., a Massachusetts-based biopharmaceutical company, to suspend all distribution of its DMD gene therapy Elevidys (delandistrogene moxeparvovec), following the deaths of three patients—two non‑ambulatory teenagers and one …
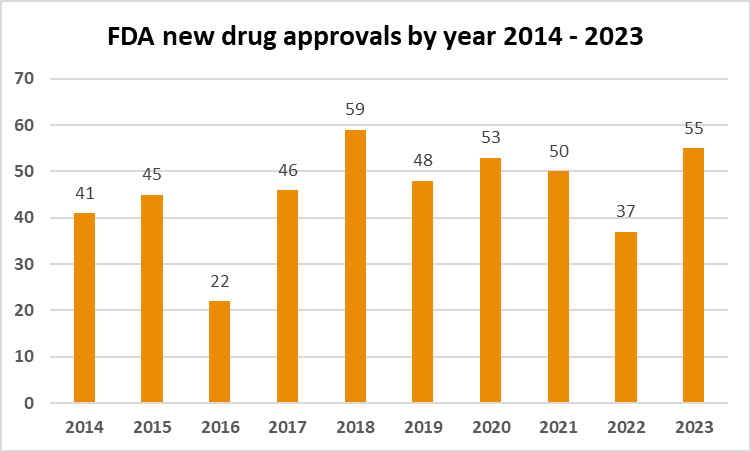
With a focus on addressing a wide spectrum of diseases and conditions, the US witnessed the most novel drug approvals in 6 years. Here we provide an overview of the key highlights in treatments …
In 2019, Novartis’s gene therapy Zolgensma made news as the „most expensive single-dose drug ever“ with $2.125 million. Just 3 years later, as of November 2022, bluebird bio’s Skysona, with the price tag of …