Written by: Adam Marťák
It will soon be 25 years since the first CDx assay came to the market paving the way for the start of the era of personalized treatment. We have discussed the last 25 years …
In 2019, Novartis’s gene therapy Zolgensma made news as the „most expensive single-dose drug ever“ with $2.125 million. Just 3 years later, as of November 2022, bluebird bio’s Skysona, with the price tag of …
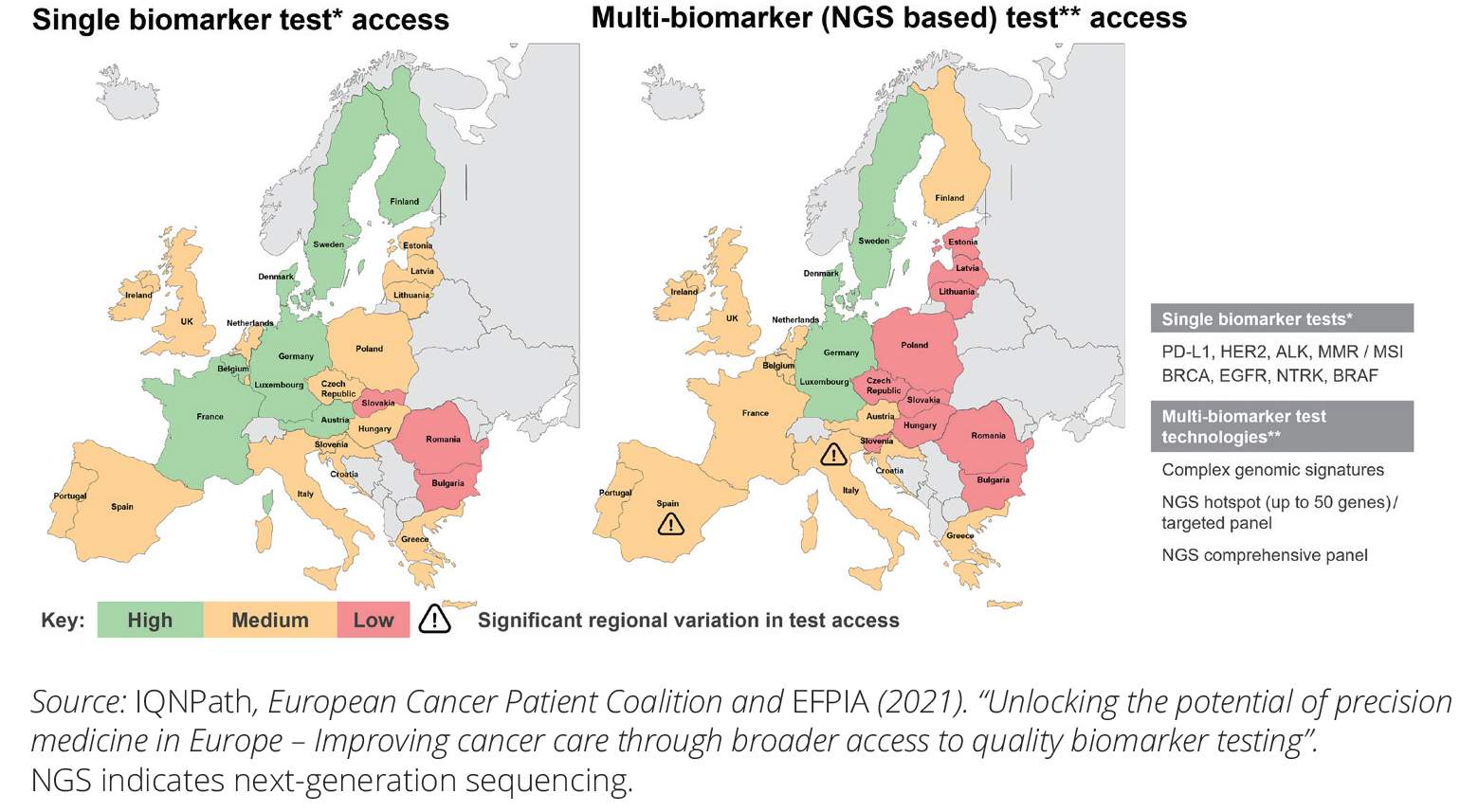
Next-generation sequencing (NGS)—a form of DNA sequencing that can examine millions of DNA molecules simultaneously—is proving to be crucial to the successful execution of the concept of precision medicine in oncology, but its utility …
The first year with eased-up COVID-19 restrictions, but with a significant economic crisis. How did the life science dealmaking fare in the first half of 2022, following the banner year of 2021? Decrease in …
The MD Regulation (MDR) 2017/745 is a recent EU directive on medical devices which entered into full force on 26th May 2021 after a transition period of three years, extended in early 2020 for …
Companion diagnostics (CDx) are used as a companion to a therapeutic agent to determine its applicability (e.g. predict response to therapy, toxicity...) to a specific person. Commonly, companion diagnostics is named as one of …
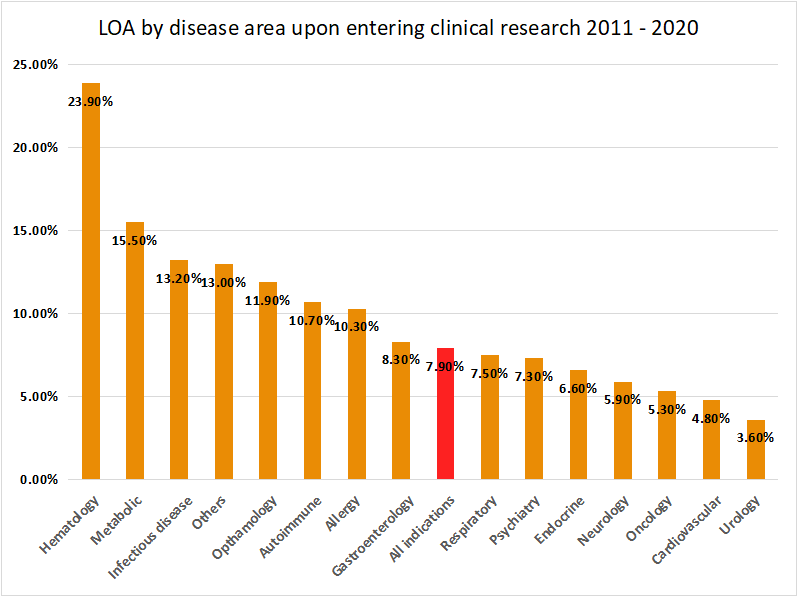
Clinical research success rates of different disease areas have far-reaching implications in the structure of corporate R&D budgets, pharmaceutical asset valuation and, eventually, M&A activity. The report published in February 2021 considers success rates of clinical …
The 2022 annual conference of the American Society of Clinical Oncology (ASCO) was held in Chicago between June 3rd-7th, widely recognised as one of the most important oncology gatherings worldwide and perhaps the most …
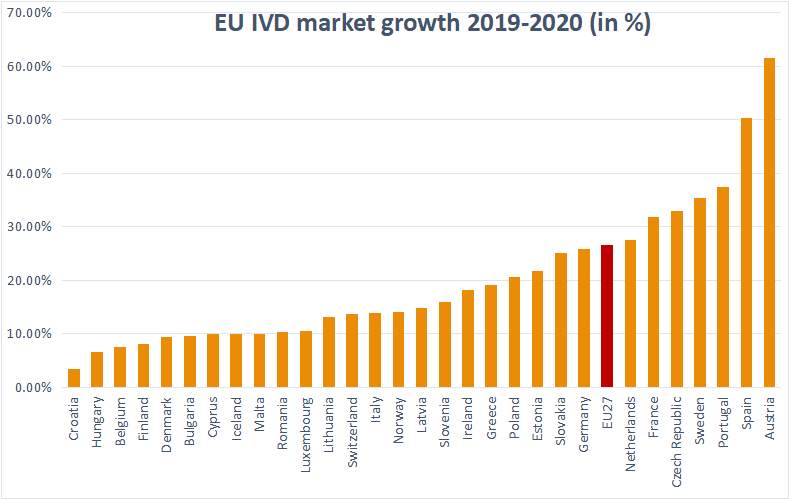
The In Vitro Diagnostic Regulation (IVDR) (EU) 2017/746 is the new EU legislation applicable to in vitro diagnostic (IVD) medical devices (shortened to IVDs here for simplicity). IVDR officially entered into force on 25th May 2017, …
The Canadian Health Technology Assessment (HTA) agency CADTH released a "Horizon Scan" identifying health tech trends set to significantly impact Canada. These trends aim to leverage data, enhance clinical workflows, and promote accessible healthcare …
The black swan event nobody could have really predicted. 400 million cases around the world, and a significant strain on healthcare systems, and the physical and mental capacities of people globally. Could there be a silver …